 Indexed in Index Medicus and Medline
Indexed in Index Medicus and Medline
HOW I DO IT
Treating male retention patients with temporary prostatic stent in a large urology group practice
Richard M. Roach, MD
Advanced Urology Institute, Oxford, Florida, USA
ROACH RM. Treating male retention patients with temporary prostatic stent in a large urology group practice. Can J Urol 2017;24(2):8776-8781.
Men with either chronic or temporary urinary retention symptoms are common patients treated in a urology practice. Both indwelling and intermittent catheterization are widely used to treat this condition. These approaches are associated with significant complications including infection and reduced quality-of-life. Infection is a target for quality improvement and cost reduction strategies in most care settings today.
We use a temporary prostatic stent (TPS) to address these issues in our practice. In this report, we describe our approach to patient selection, sizing, placement and follow up of 214 TPS placed in 56 men with chronic or temporary urinary retention in an office setting.
With the first stent placement, average indwelling time was 27 days. Thirty-two patients had multiple stents placed. Replacement was performed routinely and was generally required because underlying comorbidities precluded surgery. In these patients, an average of six stents were placed (range 2-18) with average dwell times of 31 days. Symptomatic urinary tract infections (SUTI) occurred in only 6 of 214 TPS placements (2.8%), resulting in an incident rate of 0.93 SUTI per 1,000 TPS days.
TPS is a safe and efficacious means of alleviating symptoms of urinary retention. TPS does not share the same infection risk profile or quality-of-life drawbacks associated with urinary catheters; this makes TPS use relevant as a urinary catheter alternative or when a urinary catheter is not recommended.
Key Words: urinary retention, lower urinary tract symptoms, LUTS, temporary prostatic stent, benign prostatic hyperplasia
Introduction
Male patients have a high incidence of urinary retention symptoms that increases with age.1,2 Retention symptoms occur when bladder outlet resistance exceeds the ability of the bladder to generate adequate emptying pressure.
Male retention patients often present to a urology practice with symptoms that can be described as either chronic or temporary in nature. Chronic symptoms may be related to a sustained increase in bladder outlet resistance caused by benign prostatic hyperplasia (BPH), or because of a neurogenic bladder. Similarly, temporary symptoms may be a result of edema following procedures to treat benign and malignant prostate conditions, or present in patients who suffer from postoperative urinary retention following exposure to anesthesia during non-urologic surgery.3,4
Indwelling urinary catheters (IUC) and clean intermittent catheters (CIC) are widely employed to treat patients with both chronic and temporary urinary retention. While these devices provide effective passive drainage, catheters are known to have high medical complication rates, high mortality rates, and low quality-of-life related to patient discomfort and decreased mobility.5 More recently, Umscheid and co-authors estimated 380,000 (65%-70%) catheter related urinary tract infections and 9,000 deaths could be prevented each year in the United States.6
In our transition from a volume to value-based healthcare system, one target for change has been focused on reducing use and enhancing clinical best practices for catheter care. In 2016, the U.S. Department of Health and Human Services (HHS) announced new targets for the National Action Plan to Prevent Health Care-Associated Infections with a national goal to reduce catheter associated urinary tract infections (CAUTI) 25% by 2020.7 Expenses related to hospital-acquired infections (HAI) have not been reimbursed by Medicare since 2008. Penalties for hospital acquired conditions for the 769 hospitals identified in 2016 with high rates of patient injuries represent 1% of all Medicare payments for the year and may be as high as $430 million.8
A temporary prostate stent (TPS) is an effective alternative to urinary catheters in adult male patients with chronic or temporary urinary retention symptoms. The Guideline for Prevention of CAUTIs by the Centers for Disease Control and Prevention (CDC) identifies urethral stents as an alternative to indwelling catheters and recommends further study for CAUTI prevention efficacy.9 Studies to-date have demonstrated TPS effectively drains the bladder; reducing post-void residual (PVR) while preserving continence to allow the bladder to naturally fill and empty. Because a TPS has no external components, patients often also benefit from reduced infection risk and improved quality-of-life.10-14
A recent publication examining infections after insertion of TPS in BPH patients demonstrated that when removed within 20 days of placement no bacterial colonization or infection occured.15 In a randomized trial comparing TPS to routine care, patients reported improved quality-of-life.13
Our practice utilizes TPS for treating both chronic and temporary urinary retention among our male patients. This article will explain the device, the clinical use models we’ve developed and my experience from placing 214 TPS in 56 men.
Methods and techniques
The device
The Spanner Temporary Prostatic Stent (SRS Medical, North Billerica, MA, USA) is a TPS that can be placed similarly to IUC in an office setting with only topical anesthesia. The TPS consists of a three-part system: Surveyor Urethral Measurement Device, Spanner Prostatic Stent and Introducer. The device is intended to de-obstruct the prostatic urethra for a period of up to 30 days, after which it is easily removed via the retrieval tether. Figure 1 shows the TPS de-obstructing the prostate without disrupting the external sphincter.
The TPS is anchored, tension-free, in two places; at the bladder neck by an inflatable balloon and distal to the external sphincter by a soft silicone tab. Tethers traverse the external sphincter to affix the distal anchor to the body of the stent. The external sphincter closes around these tethers, allowing the bladder to fill and empty naturally.
TPS insertion
The Introducer is inserted into the body similarly to an IUC. The proper position for TPS deployment is confirmed once the TPS balloon is inflated and gentle traction applied until resistance is felt, securing the balloon at the bladder neck. With a correctly sized device, the distal anchor is automatically positioned in the bulbar urethra as traction is applied to remove the introducer. The retrieval tether is then trimmed to length. I cut the retrieval tether short of the meatus so that there are no external components of the device. This reduces the risk of infection and eliminates the possibility that the tether is inadvertently pulled by the patient.
TPS medication
A typical medication protocol in my practice consists of antibiotics (usually ciprofloxacin 500 mg) administered at time of insertion of the TPS. Over-the-counter AZO is also used to alleviate transient dysuria and urgency symptoms caused by lower bladder outlet resistance.
TPS removal
To remove the TPS, I access the retrieval tether manually with graspers. The retrieval tether terminates in a small plug at the base of the balloon in the bladder neck. Traction on the tether deflates the balloon and allows withdrawal of the TPS.
TPS sizing
The Spanner is available in 6 device lengths (4 cm-9 cm) to accommodate different prostate lengths. Determining the correct stent size is key to device operation and patient comfort. Device size is measured from anchor to anchor, and corresponds anatomically to the distance between the bladder neck to the distal side of the external sphincter, Figure 2.
There are two ways to determine the optimal TPS size for a patient: a) the size can be calculated from a transrectal ultrasound (TRUS) measurement, or b) the size can be directly measured using a measurement device that is provided with the TPS (The Surveyor Urethral Measurement Device).
If a TRUS measurement is used, the TPS size can be calculated by taking the length from the bladder neck to the apex of the prostate in centimeters, adding 2 cms to the measurement, and then rounding up to the next cm. For example, if the prostate length is measured at 4.3 cm, 2 cm should be added to derive a device length to 6.3 cm. The TPS size would then be rounded up to a size 7.
If a direct measurement is made using the Surveyor Urethral Measurement Device, the device is introduced into the urethra and its balloon is inflated in the bladder and seated at the bladder neck. When the Surveyor is positioned in the urethra, the probe is gently advanced to detect resistance changes that indicate the location of the external sphincter. When the probe is positioned at the external sphincter, the length of the wire extending beyond the Surveyor hand piece represents the distance from the bladder neck to the distal side of the external sphincter, Figure 3.
I prefer to determine TPS size using the Surveyor Urethral Measurement Device. This technique directly accounts for balloon location at the bladder neck and length of the external sphincter. Patients who receive more than one TPS typically only need to be measured once (before initial placement). Subsequent TPS are placed without measurement.
Patient selection
We employ TPS for treating patients presenting with both chronic and temporary retention symptoms due to multiple underlying clinical conditions, Table 1.
Patients with gross hematuria or urinalysis consistent with infection or colonization are not considered good candidates for TPS. Similarly, those with known severe bladder dysfunction or incompetent sphincters are generally not offered this treatment option.
| Retention Symptom | Patient Type | Clinical Rationale | Potential Benefits |
| Chronic | BPH uncertain of detrusor function (differential diagnosis) |
Confirm detrusor function is adequate to generate void |
|
BPH unfit for treatment (concomitant conditions) |
Reduce infection risk and improve mobility |
|
|
| Temporary | BPH post-treatment (TUMT, Rezum) |
Manage LUTS related to post-procedure edema |
|
Prostate cancer post-treatment (cryotherapy) |
|||
Prostate cancer post-treatment (cryotherapy) |
Manage LUTS related to decreased detrusor pressure |
|
Results
I placed 214 TPS in 56 patients from January 2015 to December 2016. Patient ages ranged from 65 to 96 years, with mean age of 78 years. Prior to TPS placement, 39 patients reported the use of a catheter within the previous 2 weeks (31 IUC, 7 CIC). Thirteen patients reported a history of urinary tract infections. Fifteen of the 56 men subsequently underwent a deobstructing BPH procedure.
TPS were placed in an office setting, and patients were asked to perform a trial-of-void (TOV) after insertion. Cystoscopy and PVR were performed only during the first TPS sizing and placement. Cystoscopy was performed prior to TPS placement and PVR data was captured after the post-placement TOV. Because no PVR data was recorded for subsequent TPS we will describe the data from first and subsequent placements separately.
Table 2: TPS SUTI results by cohort.
Cohort |
Patients |
TPS |
SUTI |
% of TPS infection |
Avg. Indwell Days |
SUTI / 1,000 days |
First TPS |
56 |
56 |
2 |
3.5% |
27 |
1.32 |
Subsequent TPS |
32 |
158 |
4 |
2.5% |
31 |
0.68 |
All TPS |
56 |
214 |
6 |
2.8% |
30 |
0.93 |
Analysis of first TPS placement
The 56 patients with first TPS placements had an average indwell period of 27 days (range 7-84). Pre-TPS PVRs were recorded for 25 patients. Men presenting with IUC had no fluid in the bladder at the time of TPS placement and therefore PVR could not be determined. PVR in the 25 men showed an 81% decrease from pre-TPS placement (p < 0.0001), Figure 4. One additional patient had a pre-placement PVR of 995 mL was unable to generate a void after TPS placement. This patient was thought to have an atonic bladder, and was therefore eliminated from the PVR analysis.
Symptomatic urinary tract infection (SUTI) was observed in two patients receiving their first TPS, representing 3.5% of the patient cohort. One of the two infected patients reported a history of infection prior to his first TPS placement.
Analysis of subsequent TPS placements
Thirty-two patients received more than one TPS (average 6/patient, range 2-18) and averaged 31 stent indwell days per subsequent TPS placed. I routinely replace the TPS in the office at 30 days. The rationale for continued TPS use was most commonly related to underlying comorbidities and high risk for more definitive procedures.
Among subsequent TPS placements, SUTI was noted in 4 placements, representing 2.5% of the subsequent 158 TPS placed. The TPS showed a symptomatic infection rate of 0.68 infection per 1,000 indwell days. Combined, first TPS and subsequent TPS SUTI totaled 6, representing 2.8% of all TPS placed. As shown in Table 2, the SUTI incidence rate was 0.93 per 1,000 TPS days for the entire experience. This is in contrast to an estimated CAUTI incidence rate of between 3.1 and 7.5 per 1,000 catheter days in the US.9 We did not observe any negative cumulative effects in patients receiving more than one TPS.
Discussion
The mechanics of The Spanner TPS are simple, serving primarily to decrease bladder neck and prostatic resistance to assist flow while preserving continence. The 81% reduction in PVR with the first TPS placement confirms that TPS is an effective means of bladder drainage. Further, by allowing the bladder to fill and empty, TPS maintains detrusor function, which may promote bladder health among postoperative urinary retention populations and patients awaiting a BPH procedure. TPS is both safe and effective at alleviating prostate procedure-induced retention for the duration of the post-procedure healing period, long after an IUC would typically be removed.13
Our experience demonstrates a low infection rate. These data support that, if used more widely across urology offices and other care settings, TPS may be an important element in reducing complications associated with CAUTI and catalyzing reductions in prevalence, direct cost, penalties and hospital length-of-stay. While not investigated in our review, these findings may be due to an absence of external components, the natural barrier created by the closed external sphincter, and/or the benefits of allowing the bladder to fill and flush regularly.
TPS also has a profound impact on the patient’s quality-of-life, medical condition and overall health. Being able to preserve or regain normal voiding function is often life changing for men, particularly those who require use of TPS for extended periods. Though quality-of-life measures were not investigated in this review, my experience has been that men with TPS cite natural urination, increased mobility and restored sexual function high on quality-of-life benefits. When asked informally, my patients are very satisfied with the lifestyle changes and improvements they obtain from TPS.
Finally, patients without sufficient bladder or external sphincter function will fail to benefit from the advantages of TPS. Patient selection, sizing and proper placement technique are also important to maximizing the clinical experience.
Conclusions
TPS has proven a safe and efficacious means of alleviating symptoms of urinary retention across a wide spectrum of patient conditions. This is primarily because TPS does not share the same infection risk profile or quality-of-life drawbacks associated with indwelling or intermittent urinary catheters. This makes TPS use relevant both as an alternative to urinary catheters, and at times when a urinary catheter would not be recommended.
For chronic retention populations, it is a pragmatic tool for confirming detrusor function in patients transitioning to procedures, and it greatly improves the lives of patients that cannot proceed to surgery due to risk. For temporary retention, TPS is an effective tool for managing procedure-related prostatic edema, also having the ability to help reduce postoperative recovery risks through reduced infections and increased ambulation.
We have adopted TPS as a standard of care for management of male retention patients in our practice. The device is easily placed in the office, tolerated well by patients and drives improvement in several clinical parameters with few complications.
Accepted for publication February 2017
Address correspondence to Dr. Richard M. Roach, Advanced Urology Institute, 12109 CR Road, 103 Oxford, FL
34484 USA
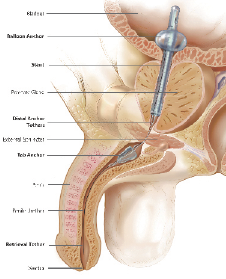
Figure 1. Sagittal plane of the male urinary tract with temporary prostatic stent (TPS) in place. A small silicone stent holds open the prostatic urethra, allowing urine to flow from the bladder, through the prostate, and beyond the external sphincter. Courtesy of SRS Medical.

Figure 2. A demonstration of temporary prostatic stent (TPS) size. Courtesy of SRS Medical.
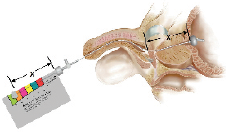
Figure 3. Temporary prostatic stent (TPS) direct sizing. Courtesy of SRS Medical.
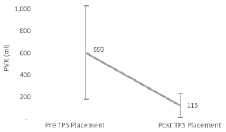
Figure 4. Patient post-void residual.
References
1. Gravas S, Bachmann A, Descazeaud A et al. Guidelines on the management of non-neurogenic male lower urinary tract symptoms (LUTS), incl. benign prostatic obstruction (BPO). 2016; http://uroweb.org/wp-content/uploads/EAU-Guidelines-Management-of-non-neurogenic-male-LUTS-2016.pdf.
2. Oelke M, Bachmann A, Descazeaud A et al. EAU guidelines on the treatment and follow-up of non-neurogenic male lower urinary tract symptoms including benign prostatic obstruction. Eur Urol 2013;64(1):118-140.
3. Locke J, Ellis W, Wallner K, Cavanagh W, Blasko J. Risk factors for acute urinary retention requiring temporary intermittent catheterization after prostate brachytherapy: a prospective study. Int J Radiat Oncol Biol Phys 2002;52(3):712-719.
4. Baldini G, Bagry H, Aprikian A, Carli F. Postoperative urinary retention: anesthetic and perioperative considerations. Anesthesiology 2009;110(5):1139-1157.
5. Saint S. Clinical and economic consequences of nosocomial catheter-related bacteriuria. Am J Infect Control 2000;28(1):68-75.
6. Umscheid CA, Mitchell MD, Doshi JA, Agarwal R, Williams K, Brennan PJ. Estimating the proportion of healthcare-associated infections that are reasonably preventable and the related mortality and costs. Infect Control Hosp Epidemiol 2011;32(2):101-114.
7. Centers for Medicare & Medicaid Services. Value Based Programs. https://www.cms.gov/Medicare/Quality-Initiatives-Patient-Assessment-Instruments/Value-Based-Programs/Value-Based-Programs.html, 2017.
8. Centers for Medicare & Medicaid Services. Fiscal Year (FY) 2016 Results for the CMS Hospital-Acquired Conditions (HAC) Reduction Program. https://www.cms.gov/Newsroom/MediaReleaseDatabase/Fact-sheets/2015-Fact-sheets-items/2015-12-10-2.html, 2017.
9. Gould CV, Umscheid CA, Agarwal RK, Kuntz G, Pegues DA, Healthcare Infection Control Practices Advisory C. Guideline for prevention of catheter-associated urinary tract infections 2009. Infect Control Hosp Epidemiol 2010;31(4):319-326.
10. Henderson A, Laing RW, Langley SE. A Spanner in the works: the use of a new temporary urethral stent to relieve bladder outflow obstruction after prostate brachytherapy. Brachytherapy 2002;1(4):211-218.
11. Corica AP, Larson BT, Sagaz A, Corica AG, Larson TR. A novel temporary prostatic stent for the relief of prostatic urethral obstruction. BJU Int 2004;93(3):346-348.
12. Grimsley SJ, Khan MH, Lennox E, Paterson PH. Experience with the spanner prostatic stent in patients unfit for surgery: an observational study. J Endourol 2007;21(9):1093-1096.
13. Shore ND, Dineen MK, Saslawsky MJ, Lumerman JH, Corica AP.
A temporary intraurethral prostatic stent relieves prostatic obstruction following transurethral microwave thermotherapy. J Urol 2007;177(3):1040-1046.
14. Dineen MK, Shore ND, Lumerman JH, Saslawsky MJ, Corica AP. Use of a temporary prostatic stent after transurethral microwave thermotherapy reduced voiding symptoms and bother without exacerbating irritative symptoms. Urology 2008;71(5):873-877.
15. Abdul-Muhsin HM, Jakob NJ, McLemore RM, McAdams SB, Humphreys MR. Infectious complications associated with the use of temporary prostatic urethral stents in patients with benign prostatic hyperplasia. Can J Urol 2016;23(5):8465-8470.
8781

