 Indexed in Index Medicus and Medline
Indexed in Index Medicus and Medline
HOW I DO IT
How I Do It: GreenLight XPS 180W photoselective vaporization of the prostate
Dean S. Elterman, MD
Division of Urology, Department of Surgery, University Health Network, University of Toronto, Toronto, Ontario, Canada
ELTERMAN DS. GreenLight XPS 180W photoselective vaporization of the prostate. Can J Urol
2015;22(3):7836-7843.
The treatments for benign prostate enlargement (BPE), also known as lower urinary tract symptoms secondary to benign prostatic hypertrophy (BPH-LUTS), have evolved significantly over recent years. Where transurethral resection of the prostate (TURP) has been the gold standard surgery for enlarged prostate glands < 80 grams, newer modalities such as laser technology have proliferated with safe and efficacious results. Notably, for prostates larger than 80-100 grams, the surgical options were an open, simple prostatectomy or perhaps a staged TURP. Both of these surgeries have the potential for bleeding complications, electrolyte abnormalities, and prolonged hospital admissions. Additional demographic and healthcare forces are also at play. Our aging population of men is being increasingly successfully treated for cardiovascular disease. This means more men are on anti-coagulation therapy, many of whom must stay on these drugs to prevent stent clotting or stroke. Hospital resources, especially overnight hospital admissions do add considerable strain to our healthcare systems. Men are also increasingly becoming more savvy consumers when it comes to their health. Many male patients would prefer to take as few medications as possible. Studies of BPH medications in Europe and the United States have shown drug discontinuation rates between 58%-70% at 1 year. Men who are faced with the choice of daily medication for life versus an outpatient procedure will often opt for the latter, which is in keeping with AUA guidelines that still put surgery as a patient choice alongside medications. Being able to offer GreenLight photoselective vaporization (GL-PVP) with the GreenLight XPS 180Watt system addresses all of these concerns. Men with bothersome BPH-LUTS with essentially any sized prostate gland, can be treated as same-day surgery requiring no overnight admission to hospital, while continuing necessary anti-coagulants, with significantly diminished risks of bleeding, erectile dysfunction, TUR-syndrome.
Just as there are many ways to perform a TURP, techniques for GL-PVP do vary. The objectives of this article are to breakdown some of the basic steps for the novice user of GL-PVP, as well as impart some “pearls” for the more experienced user. Nothing can replace hands-on experience for any surgery. The GL-PVP is unique in that there are guides such as this and previous articles, an excellent simulation device (GreenLight SIM), and mentoring programs in place. The success of many surgeries has been the standardization of the procedure. Performing GL-PVP should not be haphazard. A surgical plan based on prostate anatomy and size, cystoscopic appearance, and application of routinized techniques should yield consistent and optimal surgical outcomes.
Key Words: BPH, photoselective vaporization, GreenLight, prostate
- Introduction
- Method and technique
- The technique
- Controlling bleeding
- Unique cases
- Conclusions
- References
Overview of technology
GreenLight laser photoselective vaporization of the prostate (GL-PVP) has evolved since its early incarnation as the 80W Nd:YAG laser and potassium-titanyl-phosphate (KTP) crystal (American Medical Systems, Minnetonka, MN, USA). The subsequent generation laser was improved to 120W using a lithium-triborate (LBO) crystal, which resulted in a higher power laser, still at 532nm wavelength. The laser was emitted through a side-firing, 600-μm silica Mojo fiber.1 The current GreenLight XPS system produces up to 180W of energy using the LBO/532nm laser. Along with increased power to vaporize tissue faster, the new MoXy fiber design allows for a 50% increase in beam area (0.28 mm2 to 0.44 mm2), while keeping the same depth of vaporization (1 mm-2 mm). Therefore, the optical penetration remains the same, as does the thermal coagulation zone in all GreenLight generations. The MoXy fiber is a slightly larger 750-μm,
side-firing with 70o forward deflection laser. The Active Cooling Cap technology circulates a continuous flow of saline around the protective metal-capped fiber tip to increase fiber longevity and laser fiber protection. Enhanced software detection, called FiberLife, now protects the fiber tip from over-heating by receiving temperature feedback. When used correctly, a single MoXy fiber can now be used for an entire case, regardless of gland size. The coagulation setting now is emitted with pulses (12 Hz in 25% duty cycle, 5W to 40W) improving the lasers hemostatic
properties.2,3
Significant historical studies
Since the introduction of the GreenLight 180W XPS system in 2011 there have been increasing studies examining its safety and efficacy.4-7 Highlighted here are a couple which exemplify some of the technologies features. In February 2013, the Provincial Government of Ontario (Health Quality Ontario) came out with an Ontario Health Technology Advisory Committee (OHTAC) recommendation on photoselective vaporization of the prostate.8 The recommendations were based on a study comparing GL-PVP 120W HPS system (n = 140) to TURP (n = 24).9 Treatment outcomes were similar between the two groups based on IPSS, Qmax, SHIM, PSA, and HRQoL. The recommendations favored the use of GL-PVP as it resulted in averting 28,213 inpatient days and saved over $14 million.8 They concluded that GL-PVP is “effective, safe, and cost effective as an alternative to TURP because it can be performed in an out-patient setting, with less blood loss and a reduced need for hospital admission”.8
GL-PVP 180W XPS has also been compared to TURP in a multicenter, randomized, non-inferiority study. The GOLIATH study enrolled 291 men at 29 sites in nine European countries where the surgeons were primarily novices at GL-PVP. Prostates were < 80 cc and no patients were in retention. Primary outcomes included Qmax, post-void residual (PVR), prostate volume, and PSA. Functional questionnaires included the OABq-SF, ICIQ-SF and IIEF-5. At 12 months follow up, non-inferiority of GL-PVP 180W was maintained with no statistically significant differences between the treatment groups. Complication-free rates were similar between GL-PVP and TURP group (84.6% and 80.5% respectively). Rates of unresolved urinary incontinence at 1 year were equal, with four patients in each group.10
As residents we are taught that every surgery has a preparatory phase of investigations and decision making, followed by careful planning and execution of a series of fairly sequential steps. While some latitude is given for adapting to real world situations, orderliness to surgery is critical to its safe and effective completion. GL-PVP should be no different. I have adapted aspects of standard TURP, GL-PVP vaporization-resection technique,11,12 GL-PVP enucleation,13 GL-PVP vaporization-incision technique14 and standard GL-PVP15 cases to come up with a standard approach to patient evaluation, operative set up, surgical technique and postoperative care that can be applied to nearly every case. Following these guidelines has allowed new users of GL-PVP to more quickly learn the key steps and avoid the discouragement many novices experience when things don’t go exactly according to plan.
Patient selection and work up
The criteria for offering men surgical management for BPH-LUTS remain unchanged and are consistent with the Canadian Urological Association, American Urological Association, and European Urological Association guidelines.16-18 Subjective complaints of bothersome LUTS coupled with objective findings on uroflowmetry, for example, are often the initiator of treatment. Validated questionnaires such as the IPSS or AUA-Symptom Index serve as confirmation as well as baseline measurements to determine progression of symptoms or improvement with treatment.17 The shared decision to move forward with surgery remains contingent on patient response to behavioral or medical management, as well as patient preference. I find increasingly men wishing to have GL-PVP as an alternative to lifelong medical therapy because of it’s decreased morbidity and out—patient setting.
The preoperative evaluation for every patient includes urinalysis, cystoscopy with uroflowmetry and PVR urine ultrasound, as well as a transrectal ultrasound (TRUS). One wants to make sure there is no other lower GU tract pathology, such as stricture, bladder stone, or malignancy. Also use this as an opportunity to plan the surgery. Assess the prostatic urethral length and the anatomic features of the lateral and median lobes. Are the lobes kissing? How large of an intravesical component is there? Is there a large ball-valve median lobe? How far away are the ureteric orifices from the bladder neck on retroflexion? These features may modify your approach during the case but are still part of a standardized algorithm. TRUS determination of gland size is important for surgical scheduling and patient selection. There is a fairly consistent relationship between prostate size and vaporization time.19 Knowing the size and characteristics of the prostate will help plan the case, especially the first 10 or so cases when patient selection is important for confidence building and outcomes. Prostates between 40 cc-80 cc without a large median lobe and not on anti-coagulation are good cases for novices to practice the standardized approach.
Laser fiber overview
The novice user should understand that GL-PVP is a two-handed, dynamic operation. This means that while the dominant hand moving the laser fiber forward and back, as well as twisting it side to side to vaporize tissue, the opposite hand is moving the camera forward and back (proximal and distal) to maintain visibility and lens-to-fiber tip distance. A note about lens-to-fiber tip distance: there is a small ridge half way along the metal cap circumferentially. To avoid burning your scope lens, always keep that ridge visible (i.e. never bring the fiber in so close to the lens that you cannot see the ridge).
Understanding the basics of laser properties will make you a better operator. The laser beam is side-firing and 70o forward. The optical depth of penetration is 0.8 mm with heat penetrating an additional 1 mm-2 mm. The optimal tissue-laser fiber distance is the same as one fiber cap width (1.8 mm), so staying just off the tissue will give the best vaporization results. The laser loses efficiency if you’re too far away from tissue therefore you want the laser aimed at tissue no more than 3 mm away. This matters because your sweep speed, tissue distance, and sweep width will be guided by these principles.
The bubbles created can also assess the distance to the tissue and efficiency of vaporization. Large size “scuba” bubbles are good, while fine “champagne” bubbles mean the fiber is too far away from the tissue. If you drag the fiber tip along the tissue, the heated tissue will become adherent to the lens inhibiting laser emission. The beam will lose its columniation and power, eventually requiring the use of a new fiber. If your sweeps are too wide (> 30 degrees), your laser beam will shoot off into dead-space (not onto tissue) and be inefficiently used. If you sweep too quickly, the tissue doesn’t heat enough resulting in coagulation/charring (doesn’t reach 100 oC).20 The tissue will appear desiccated and may result in worse dysuria for the patient. Sweep speed should be set like a metronome. Approximately 2 seconds per sweep of 30 degrees rotation results in efficient vaporization.21
Another tip for the GL-PVP user is how to cover completely the surface area of the prostate for vaporization. In a TURP, one picks a start and end point of where the resection loop enters and exits the tissue. In a GL-PVP one similarly need to imagine discreet strips of tissue from the bladder side back towards the verumontanum. The analogy I use when teaching is farming, particularly a terraced rice field. You need to appreciate the linear rows of tissue to vaporize along with the topography of the tissue. The goal is to sequentially move from one side to another (i.e. midline to lateral), moving forward and back along the rows of tissue that you have visually created. Eventually everything should become flattened out equally. The next important part is how your sweeps vaporize the tissue as you rotate the fiber. The analogy I use is painting a wall with a roller brush. When you paint a wall one starts at one point moving the brush up then down, moving sideways just enough that the paint barely overlaps. This covers the wall completely and efficiently. The same is true for laser vaporization. If you move the laser and/or camera too quickly, you won’t cover all the surface area. It will look like you’re painting a series of the letter “W” (i.e. WWWW). Remember to “always be moving”. Your fiber should continuously be sweeping side to side, while the same hand can move it in and out of the scope. The opposite hand can be moving the camera. If you stop sweeping while firing the laser, you will drill a hole and potentially start bleeding. A final note on this topic: stick to your plan (divided terraced rice field covered like your painting a wall). Be methodical. It’s very tempting to be opportunistic and move from floor to right lateral to left roof to left base, as you see tissue seemingly appear. Instead, make a mental note of where you will eventually need to vaporize. One exception is when some tissue is obscuring your view of the area beyond which you are trying to vaporize.
The goal of this technique is to standardize a general sequence of steps that can be used by the novice and advanced user alike. It is applicable to nearly every shape, size, and configuration of prostate gland. Whether I’m doing a 40 cc or 240 cc prostate, my approach remains the same. This way you can always know where you are in the case (time), where you are inside the prostate (location), what still needs to be done (progress).
Scope insertion and preparation
After my equipment has been checked and set up I proceed with inserting the Storz 24F continuous flow resectoscope with a laser bridge. Entry with an obturator technique is fine but do not force it as bleeding at the start of the case can make visibility at the outset challenging. If in doubt, enter under direct vision. The bladder is drained of urine and cystoscopy carried out. It is at this point, once I’m safely in the bladder with no urethral complication, that I have the nurse open the MoXy laser fiber box. While looking for the ureteric orifices, avoid rubbing the median lobe/bladder neck with the scope to avoid bleeding. I assess the size and shape of the median lobe and pull the scope back to the verumontanum, calculating the prostate length. Before I begin vaporizing, I check the patients’ blood pressure with the anesthesiologists. I’ve already discussed prior to induction or spinal that, if safe for the patient, the target blood pressure throughout the case is a systolic BP of 100-110 mmHg. An optional step now is to mark out the distal vaporization border using the coagulation circumferentially, just a centimeter or so proximal to the verumontanum. I keep the fiber tip a bit further from the tissue, in the center of lumen, to prevent bleeding. You just want to blanche the mucosa using the standard power setting of 35W coagulation. Remember, the external sphincter is omega-shaped and more proximal anteriorly. You might avoid this marking step if the mucosa looks particularly friable or vascular.
Creation of lateral grooves to capsule, Figure 1b
With the prostate assessed and marked out, one should have a starting surgical plan in place. The power settings at the start of the case should be 80W vaporize and 35W coagulate. These first initial steps to follow are all “set up”. They set the parameters for where you will ultimately vaporize on higher power. There is no need to change the coagulation setting under any circumstance. All the mucosa lining the prostatic urethra should be vaporized on 80W. This will minimize bleeding. Once the mucosa is stripped away, one can increase energy incrementally assessing the tissues response and whether is bleeds. The underlying adenoma can be efficiently vaporized on 140W-180W but remember to be patient and remove the mucosa on low power and work your way up. Assuming there is a normal median lobe and not enormous, begin by identifying the intersection of the median and lateral lobe at the bladder neck. Begin here, vaporizing from the bladder neck at the 4:00 o’clock position straight back towards the verumontanum, though I only go part/half way back on large prostates. The goal is to make a deep groove out towards the capsule. This sets my depth of vaporization. The longitudinal incision or groove has gone deep enough when the fibers begin to look capsular and they don’t vaporize as easily. Also I look forward at the bladder edge of the groove to see when it become flush to the bladder wall. At this point I tilt my laser up towards 2:00 o’clock and incise the prostate away from the bladder wall edge. I repeat the same deep groove at the contralateral 8:00 o’clock position. The lateral grooves are created using lower 80W-100W vaporize setting to prevent bleeding and capsular perforation.
Creation of vertical grooves downward, Figure 1c
and 1d
The next move is the creation of deep grooves on either side of the median lobe. At the medial aspect of the lateral groove, just beside the lateral top edge of median lobe, the laser is aimed downward. Using 80W and possibly increasing to 100W, a wide groove is created. I keep going until the depth of the groove is flush to the bladder floor, assessed by looking over the edge of my groove into the bladder, where I see trigone straight ahead. This is repeated on opposite side of median lobe. I now have a mound of median lobe, which can be cleared away on higher energy. When done this way, initially small appearing prostates can actually show themselves to be quite large and obstructing, much like the tip of an iceberg. Approach the median lobe mound from the sides, aiming the laser horizontally and moving the scope up and down like painting a wall. The visual description would be moving up and down the side of a cliff. I also alternate sides, approaching the median lobe from left and right. Avoid treating the base too much, as you’ll get a mushroom-on-a-stalk piece of tissue, which is trickier to treat. One should take care at the bladder neck to avoid aberrant lasing into the bladder that could injure a ureteric orifice. At the end I flatten everything by lasing nearly straight down.
Vaporizing anterior-lateral tissue, Figure 1e
When the initial lateral grooves are created at 4 o’clock and 8 o’clock, the overhanging antero-lateral tissue above the groove must be vaporized. Aiming the laser up from underneath and the side can quickly and efficiently vaporize this tissue on high power (140W-180W).
Vaporizing inferior-lateral tissue, Figure 1e
The tissue beneath the initial lateral grooves, between the 4 o’clock/8 o’clock and 6 o’clock grooves can now be vaporized on higher power (140W-180W). By making that first lateral groove, the incision sets the limit of how much vaporization is required. It removes the uncertainty of how far out to vaporize. One wants to incise far enough distally, towards the verumontanum, so that a continuous cavity is created from lateral capsule along floor where median lobe used to sit and up to contralateral capsule. At this point a large TUR-like defect in the half of the prostate closest to bladder neck has been formed. What remains are the lateral lobes extending from approximately the mid-gland back towards the verumontanum.
Superior and inferior division of lateral lobes, Figure 2b, 2c and 2d
Creating a working channel: In order to vaporize tissue with good visibility, space needs to be created away from the tissue. At this point reassess the distance to the verumontanum and move inward to the cut edge of the lateral lobes (where the vaporization ended). Turn the energy back down to 80W as one will be vaporizing mucosa and want to prevent bleeding. I divide in my mind the lateral lobes into a superior and inferior half, much like the northern and southern hemisphere of a globe. I bring my scope up above the horizontal midpoint, as I want to create a working space superiorly first. Use the distal end of the scope to push the contralateral lobe lateral, giving oneself a window of space to put out the laser fiber and begin vaporizing the ipsilateral lobe. Work up and down the top half of the lobe removing the mucosa and simultaneously drawing the scope back toward the apex where one will want to stop proximal to the verumontanum. Bleeding can be encountered here, particularly at the cut edge of the mucosa. Try to keep moving to complete as much mucosal removal as possible, since once you begin to vaporize the underlying adenoma your scope will compress the bleeding on the distal cut edge. Once a small space is created on the opposite lateral lobe with the mucosa removed, I begin to vaporize the superior half of the lateral lobes. The power is incrementally increased (120W-180W) while the upper half of the lateral lobes are vaporized until they are flush with the TUR-like defect created in the bladder neck-half of the prostate. Again, by making those initial lateral deep grooves, the lateral border of vaporization has been set and it’s now a matter of connecting the topography of the adenoma back to the capsule. Once this is completed, a horizontal shelf of tissue corresponding to the surface of the inferior half of the lateral lobes closest to the verumontanum remains. This can quickly be vaporized straight downward until these lobes have been removed. Again the medial aspect of the lobes and the area closest to the apex should be vaporized on 80W when removing the mucosa and nearing the sphincter complex. I keep the same technique of sweeping 30 degrees with 2-second sweeps.
Check the anterior tissue
A fairly wide-open TUR-like defect has been created. With the scope at the verumontanum, look up anteriorly to see what tissue is hanging down. Bleeding from the roof can be difficult to see and control so turn the energy down to 100W-140W. Position the scope as distal as possible such that the scope beak does not lift up tissue that should be vaporized. I don’t believe the anterior tissue to play a large component of urodynamic obstruction therefore it is not vital to vaporize aggressively between 10 o’clock and
2 o’clock.
Case conclusion
With the last apical and lateral tissue vaporized I am now ready to assess the TUR-like defect. With the in-flow turned off, look for bleeding. I typically don’t worry about a small amount of venous ooze. I sometimes coagulate from a distance (2 cm) above the tissue to create a charred clot which tamponades the ooze. If tissue falls down from above with the irrigant turned off, vaporize it away. Check the bladder and ureteric orifices for injury. Lastly assess the bladder neck to determine if the catheter with pass easily or if it should be inserted with a stylet. The bladder is left full and the scope is removed. I use a regular 22F 3-way catheter, with an optional stylet introducer. I fill the balloon with 30 cc sterile water and place the catheter on traction if there is any hematuria. My traction technique is quite simple; a single 4 x 4 gauze unfolded and wet. Tied around the catheter with a simple knot. I pull the catheter on traction and slide the knot up to the meatus where it holds the catheter on traction. It’s removed after an hour in the recovery room. Continuous bladder irrigation is optional but seldom necessary. Surgeries performed in the morning with general anesthesia can have an active voiding trail prior to discharge (fill with 250 cc-300 cc water). Those done in the afternoon, done with spinal anesthesia or who had prior retention go home with their catheter, where a visiting nurse removes it the following day at home. I only admit patients overnight for medical comorbidity issues if required, otherwise every GL-PVP, regardless of anticoagulation status or gland size, is discharged home the same day.
The most challenging and frustrating aspect of GL-PVP can be hemostasis. The GL-PVP allows the operator to work the vast majority of time in a bloodless field. Occasionally, a small vessel will begin to bleed and knowing how to use the TruCoag setting, MoXy fiber metal cap, and the scope beak will make these situations manageable. Understand that a bleeding vessel has a circular shape to it. Shooting a laser beam directly into it will actually make the bleeding vessel bigger. The first step to control bleeding is to get right up close to the bleeding vessel. The irrigation will clear the view and the open vessel should fill the screen image. If tissue is obscuring the view of the bleeder, vaporize around it to make it visible. Next, use the beak of the scope or the tip of the fiber to compress the vessel from circular to oblong/oval, like pinching it shut. Aim the coagulation beam around the edges of the bleeding vessel and move the beam circumferentially around the bleeder, like sealing the edges shut. Aim the beam obliquely at the vessel, never directly into the bleeding. Coagulation is delivered in short bursts of a few seconds, followed by reassessment to see if bleeding has been controlled. The entire scope can be used to compress a larger bleeding area. A degree of patience and calm must be maintained to manage bleeding. In a worst case, a bugbee electrode or bipolar loop can be inserted to manage the large bleeder. A note on the anti-coagulated patient: certainly if patients can safely stop their blood thinners then have them do so. GL-PVP can safely be performed in fully, irreversibly anti-coagulated patients. Extra care should be taken and lower energy setting should be used throughout the case. I tell my patients to resume their anti-coagulation medications the day after surgery or once their urine is clear.
Lateral lobes only
There occasionally is a prostate that has only very large lateral lobes with no median lobe. While a straightforward vaporization is possible, a variation of the above-described technique can be employed. I come up two-thirds along the lateral lobe at the bladder neck and make a lateral deep groove (as described above). I keep creating that long incision laterally for 3 cm-5 cm or until I reach capsule. At this point I aim my fiber downward and make the deep vertical groove downward (like I would on the lateral edges of the median lobe). I go down until I’m flush with the bladder neck. In essence, I have created a median lobe out of the lateral lobe. I then vaporize it in the same fashion as the median lobe.
Large median lobe
The very large or very high or enormous median lobe can be difficult to treat; especially when they’re so big the scope and laser barely reach their apex. The trick is to bisect this type of large median lobe. Start at the top and create a deep groove right down the middle, splitting it into two halves. Then proceed with the deep vertical grooves on either side of the median lobe, effectively creating two mounds to vaporize until flush to the bladder base. Always keep in mind the location of the ureteric orifices. In large glands with protruding median lobes, they are often tucked underneath the lobe and can be easily injured.
Preserving ejaculation
Attempts of ejaculation-preservation can be performed with modifications to the standard GL-PVP technique though these modifications are based on anecdote and expert opinion. Keeping flaps of apical tissue, preserving the precollicular tissue, and paracollicular prostate tissue (ejaculatory hood) may diminish rates of retrograde ejaculation, though I always counsel and consent patients to expect retrograde ejaculation. It’s not something that can be guaranteed no matter what the technique.
GL-PVP has been shown to be effective for treating a wide range of prostate glands, with the benefit of maintaining anti-coagulation while minimizing intraoperative blood loss and electrolyte disturbances from fluid absorption. The technology provides a significant cost savings to healthcare systems by avoiding in-patient hospital admissions. However, all of these benefits can only be realized if surgeons are comfortable and adequately trained on this technology. The web-based accreditation programs along with simulation use or mentoring when available are excellent ways to learn the basics. Proper patient evaluation and selection will allow the new user to get comfortable with the laser-to-tissue interaction and vaporization techniques. Above all else, a standardized approach to GL-PVP must be followed to ensure safety, efficiency and success. Just as I learned my mentors’ ways of approaching cases, you can adapt the steps described herein. I believe a standardized technique, such as this, should be easily reproducible. These steps can be applied to nearly any prostate to ensure a consistent successful outcome. Like everything that is new, take your time, build your experience and confidence, learn from mistakes and refine your technique.
Accepted for publication May 2015
Acknowledgement
We wish to thank illustrator Michael Corrin, Lecturer, Biomedical Communications, Department of Biology, University of Toronto, Ontario, Canada
Address correspondence to Dr. Dean S. Elterman, Toronto Western Hospital, Main Pavilion, 8th Floor, Room 317, 399 Bathurst Street, Toronto, ON M5T 2S8 Canada
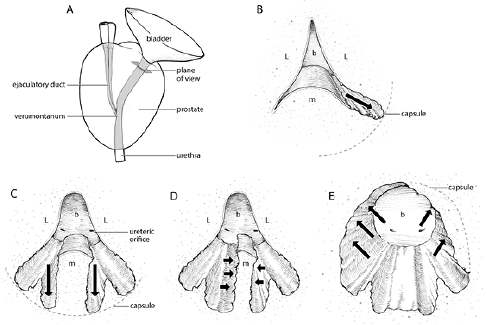
Figure 1. A) Plane of view of prostate illustrations B through E. B) Creation of lateral grooves to capsule. Start at intersection of median and lateral lobe at bladder neck. Vapor-incise on 80W longitudinally back toward verumontanum creating deep grooves out towards capsule at 4 o’clock and 8 o’clock positions. C) Creation of vertical grooves downward. At lateral edge of median lobe create wide, deep grooves straight down at 6 o’clock towards floor of bladder.
D) Vaporize median lobe. Approach tissue laterally from within the created grooves or obliquely from above using high energy (140W-180W). E) Vaporize anterior-lateral and inferior-lateral tissue. The remaining tissue between the created grooves is vaporized on high energy (140W-180W)
L = lateral lobe; M = median lobe; B = bladder
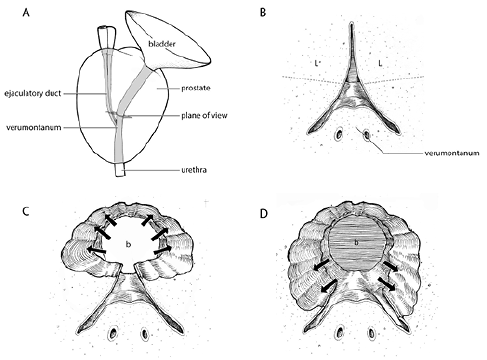
Figure 2. A) Plane of view of prostate illustrations B through D. B) Superior and inferior division of lateral lobes. Visually divide the lateral lobes closest to the verumontanum into superior (upper) and inferior (lower) halves. C) Create a superior working channel. Begin by vaporizing the superior half of the lateral lobes initially. Start in low energy (80W) and increase to higher energy (140W-180W) once mucosa removed back to near verumontanum demarcated with coagulation at start of case. Once completed, a horizontal shelf of tissue corresponding to the surface of the inferior half of the lateral lobes closest to the verumontanum remains. D) Vaporization of inferior lateral lobes. The remaining shelf of tissue inferiorly is vaporized away, creating a TUR-like defect contiguous from the bladder neck to the verumontanum.
1. Gu X, Strom K, Spaliviero M, Wong C. Does prostate configuration affect the efficacy and safety of GreenLight HPS laser photoselective vaporization prostatectomy (PVP)? Lasers Med Sci 2013;28(2):473-478.
2. Emara AM, Barber NJ. The continuous evolution of the Greenlight laser; the XPS generator and the MoXy laser fiber, expanding the indications for photoselective vaporization of the prostate. J Endourol 2014;28(1):73-78.
3. Malek RS, Kang HW, Peng YS, Stinson D, Beck MT, Koullick E. Photoselective vaporization prostatectomy: experience with a novel 180 W 532 nm lithium triborate laser and fiber delivery system in living dogs. J Urol 2011;185(2):712-718.
4. Hueber PA, Bienz MN, Valdivieso R et al. Greenlight photoslective vaporization of the prostate (PVP) for benign prostatic hyperplasia using the 180W-XPS system: A multicenter international study of the impact of prostate size on safety and outcomes. J Urol 2015 Apr 4. Epub ahead of print.
5. Eken A, Soyupak B, Acil M, Arpaci T, Akbas T. Safety, efficacy and outcomes of the new GreenLight XPS 180W laser system compared to the GreenLight HPS 120W system for the treatment of benign prostatic hyperplasia in a prospective nonrandomized single-centre study. Can Urol Assoc J 2015;9(1-2):e56-e60.
6. Brunken C, Seitz C, Woo HH. A systematic review of experience of 180W XPS GreenLight laser vaporization of the prostate in 1640 men. BJU Int 2014 Oct 13. Epub ahead of print.
7. Altay B, Erkurt B, Kiremit MC, Guzelburc V, Boz MY, Albayrak S.
180-W XPS GreenLight laser vaporization for benign prostate hyperplasia: 12-month safety and efficacy results for glands larger than 80 mL. Lasers Med Sci 2015;30(1):317-323.
8. Bowen JM, Whelan JP, Hopkins RB et al. Photoselective vaporization for the treatment of benign prostatic hyperplasia. Ont Health Technol Assess Ser 2013;13(2):1-34.
9. Whelan JP, Bowen JM, Burke N et al. A prospective trial of GreenLight PVP (HPS120) versus transurethral resection of the prostate in the treatment of lower urinary tract symptoms in Ontario, Canada. Can Urol Assoc J 2013;7(9-10):335-341.
10. Bachmann A, Tubaro A, Barber N et al. A European multicenter randomized noninferiority trial comparing 180 W GreenLight XPS laser vaporization and transurethral resection of the prostate for the treatment of benign prostatic obstruction: 12-month results of the GOLIATH study. J Urol 2015;193(2):570-578.
11. Son H, Ro YK, Min SH, Choo MS, Kim JK, Lee CJ. Modified vaporization-resection for photoselective vaporization of the prostate using a GreenLight high-performance system 120-W Laser: the Seoul technique. Urology 2011;77(2):427-432.
12. Elterman DS, Chughtai B, Lee R et al. Comparison of techniques for transurethral laser prostatectomy: standard photoselective vaporization of the prostate versus transurethral laser enucleation of the prostate. J Endourol 2013;27(6):751-755.
13. Gomez Sancha F, Rivera VC, Georgiev G, Botsevski A, Kotsev J,
Herrmann T. Common trend: move to enucleation-Is there a case for GreenLight enucleation? Development and description of the technique. World J Urol 2015;33(4):539-547.
14. Ben-Zvi T, Hueber PA, Abdollah F et al. Short term outcomes of GreenLight vapor incision technique (VIT) of the prostate: comparison of outcomes to standard GreenLight 120W HPS vaporization in prostate volumes greater than 80 cc. Can J Urol 2013;
20(1):6633-6639.
15. Malek RS, Kuntzman RS, Barrett DM. High power potassium-titanyl-phosphate laser vaporization prostatectomy. J Urol 2000;
163(6):1730-1733.
16. Nickel JC, Herschorn S, Corcos J et al. Canadian guidelines for the management of benign prostatic hyperplasia. Can J Urol 2005;
12(3):2677-2683.
17. McVary KT, Roehrborn CG, Avins AL et al. Update on AUA guideline on the management of benign prostatic hyperplasia. J Urol 2011;185(5):1793-1803.
18. Madersbacher S, Alivizatos G, Nordling J, Sanz CR, Emberton M,
de la Rosette JJ. EAU 2004 guidelines on assessment, therapy and follow-up of men with lower urinary tract symptoms suggestive of benign prostatic obstruction (BPH guidelines). Eur Urol 2004;46(5):547-554.
19. Ben-Zvi T, Hueber PA, Liberman D, Valdivieso R, Zorn KC. GreenLight XPS 180W vs HPS 120W laser therapy for benign prostate hyperplasia: a prospective comparative analysis after 200 cases in a single-center study. Urology 2013;81(4):853-858.
20. Osterberg EC, Kauffman EC, Kang HW, Koullick E, Choi BB. Optimal laser fiber rotational movement during photoselective vaporization of the prostate in a bovine ex-vivo animal model. J Endourol 2011;25(7):1209-1215.
21. Kauffman EC, Kang HW, Choi BB. The effect of laser-fiber sweeping speed on the efficiency of photoselective vaporization of the prostate in an ex vivo bovine model. J Endourol 2009;23(9):
1429-1435.
7843

