 Indexed in Index Medicus and Medline
Indexed in Index Medicus and Medline
Free Articles
HOWIDOIT
Functional, oncologic, and technical outcomes after endoscopic groin dissection for penile carcinoma
Daniel J. Canter, MD,1 Ryan W. Dobbs, MD,1 S. Mohammed A. Jafri, MD,1 Lindsey A. Herrel, MD,1 Kenneth Ogan, MD,1 Keith A. Delman, MD,2
Viraj A. Master, MD1
1Department of Urology, Winship Cancer Institute, Emory University School of Medicine, Atlanta, Georgia, USA
2Department of Surgical Oncology, Winship Cancer Institute, Emory University School of Medicine, Atlanta, Georgia, USA
CANTER DJ, DOBBS RW, JAFRI SMA, HERREL LA, OGAN K, DELMAN KA, MASTER VA. Functional, oncologic, and technical outcomes after endoscopic groin dissection for penile carcinoma. Can J Urol 2012;
19(4):6395-6400.
Penile cancer is a rare cutaneous malignancy that frequently spreads to the regional inguinal lymph nodes with a prolonged locoregional phase. An inguinal lymph node dissection may be both diagnostic and therapeutic, even in the setting of advanced disease. Despite its proven oncologic importance and efficacy, an inguinal lymphadenectomy remains underutilized, even with the publication of guidelines advocating its use. Failure to apply this modality is most likely due to the significant morbidity associated with a traditional open approach, including flap necrosis, wound infection and debilitating lymphedema.
The risks and complications associated with an open inguinal lymph node dissection have driven several investigators to develop techniques for performing a minimally invasive endoscopic inguinal lymph node dissection that is oncologically equivalent to the ‘gold standard’ open approach, while potentially minimizing the complications traditionally seen with the open technique. In this report, we detail our technique for performing a minimally invasive endoscopic groin dissection with inguinal lymphadenectomy for penile carcinoma. We also present preliminary complication and short term oncologic data employing this surgical technique in an initial cohort of patients.
Key Words: penile carcinoma, laparoscopy
- Introduction
- Methods
- Pre-procedure instructions
- Surgical technique
- Post-procedure instructions
- Results
- Discussion
- Conclusions
- References
Penile cancer is a rare malignancy accounting for an estimated 1570 new cases and 310 deaths in the United States in 2012.1 Despite the low incidence, each case potentially carries significant morbidity and mortality as well as unquantifiable quality of life issues related to self-image issues surrounding penile surgery. Penile cancer spreads in a predictable fashion to the regional lymph nodes prior to hematological (distant) dissemination. Lymph node (LN) metastases are a common pathological event in the progression of penile cancer, demonstrating varying rates dependent on the pathologic stage of the primary tumor-- 50% with pathologic Ta/T1 disease and 29%-64% with pathologic T3/T4 disease.2
Current imaging modalities have varying abilities to detect clinically occult lymph node disease as 20%-25% of penile cancer patients determined to be clinically node negative have pathologically involved lymph nodes at the time of presentation.3 The presence and degree of involvement of regional lymph nodes are the most important prognostic factors for a patient with penile cancer.3 Fortunately, despite the presence of positive regional lymph nodes, only 1%-2% of patients will initially present with distant metastases3 due to the fact that penile cancer exhibits a prolonged locoregional phase. This biological behavior provides an opportunity for surgical intervention in the form of inguinal lymphadenectomy to effect a durable cure. Despite the evidence suggesting improved oncologic outcomes for early and extensive lymphadenectomy for patients with penile cancer,4,5 the approach is not regularly utilized when despite being indicated,5 most likely due to the significant morbidity of the traditional open approach. In an attempt to minimize the morbidity associated with inguinal lymphadenectomy, we have recently adapted and continue to prospectively evaluate in a randomized clinical trial an endoscopic approach to performing an inguinal lymph node dissection (clinicaltrials.gov identifier NCT01526486).6 Although not yet complete, this prospective trial has initial promising results. In the present report, we detail our technique for performing a minimally invasive endoscopic groin dissection with inguinal lymphadenectomy for penile carcinoma. We also present our initial complication and short term oncologic data employing this surgical approach.
At Emory University Hospital, endoscopic groin dissection was first performed in August 2008. From August 2008 to the present, a total of 10 patients underwent 19 groin dissections for squamous cell carcinoma of the penis. Indications for surgery for patients with penile cancer were palpable lymph nodes at diagnosis or the presence of high risk features, defined as pathologic stage ≥ T2, high grade histology, and/or lymphovascular invasion of the primary tumor. We also perform this procedure as needed in patients with scrotal carcinoma or melanoma. Informed consent was obtained from each patient, specifically mentioning the novelty of the procedure as well as the possibility of unanticipated complications. Additionally, Institutional Review Board (IRB) approval was obtained prior to performing any endoscopic groin dissections.
Data was reviewed from a prospectively maintained institutional database. The type of complications were also recorded in a prospective fashion and were stratified as minor or major based on the Clavien-Dindo classification system7 as well as the definitions published by Bevan-Thomas.8 According to the Bevan-Thomas definitions, major complications included death, severe leg edema interfering with ambulation, skin flap necrosis, re-hospitalization for wound care, wound infection require intravenous antibiotics, treatment of sepsis, deep venous thrombosis or re-exploration/other invasive procedures performed in the operating room (e.g. incision and drainage of abscess or seroma). Minor complications included conditions requiring local wound debridement in the clinic, mild (trace 1+ pitting) to moderate (2+ pitting) leg edema, seroma formation not requiring aspiration and minimal skin edge necrosis requiring no therapy. Patients with both major and minor complications were classified in the major complication group. Patients with more than one minor complication were classified in the minor complication group. Early oncologic outcomes that were measured in the penile cancer group included local recurrence, development of metastatic disease, and mortality.
1) Appropriate patients undergo a thorough preoperative counseling session, discussing the risks, benefits, and alternatives to performing endoscopic groin dissection.
2) Anticoagulant and antiplatelet agents should be discontinued at the appropriate time before the procedure, typically 5-7 days prior to the lymphadenectomy.
1) Patients receive intravenous antibiotics effective against skin microbes--usually penicillin, a third-generation cephalosporin, or vancomycin in penicillin-allergic patients--1 hour prior to the beginning of surgery.
2) On a split-leg table, the surgical assistant stands on the lateral side of the leg being dissected, and the surgeon stands in between the patient’s legs, Figure 1a. The supine patient is positioned in a frog-leg position on a split-leg table with their knees supported by small towels, Figure 1b. Care should be taken so that the knee supports do not obstruct port placement, especially medially.
3) Prior to positioning, the boundaries of the femoral triangle are marked. The base of the triangle is a line drawn from the anterior superior iliac spine to the pubic tubercle along the course of the inguinal ligament. The lateral boundary is the sartorius muscle angling toward the apex. The medial boundary is the adductor longus muscle, which also angles towards the apex. This inverted triangle guides correct port placement and delineates the extent of the inguinal dissection. Suprapubic skin should be prepared into the field to monitor for the development of intraoperative crepitus. A safety belt is placed tightly on the abdomen which may aid in decreasing cephalad migration of CO2 through the subcutaneous tissues.
4) Initial trocar placement is made 3 cm inferior to the apex of the femoral triangle with a 12 mm incision is made through skin and Camper’s fascia. During placement of this apical port, it is important to identify Scarpa’s fascia, which can reliably be identified by its white layer. Once Scarpa’s fascia is identified, a finger should be used to bluntly develop a potential space above Scarpa’s fascia of at least 5 cm on either side of the initial skin incision, Figure 2. The initial finger dissection is a critical step to rapidly and safely establish a working space with minimal blood loss. One of the initial observations we made as we have gained more experience with this procedure is that the skin flap can be made thin without risking skin necrosis. Initially, we were probably too timid in creating our working space and may have compromised our nodal yield.
5) Once the proper plane has been developed, a 12 mm Origin balloon port (Origin Medsystems Inc, Menlo Park CA, USA) is placed and the working space is created by inflating the balloon to 25 mm Hg of pressure for 10 minutes. High pressure insufflation of carbon dioxide into the subcutaneous space helps to rapidly establish this working space.
6) A zero-degree 10 mm optic laparoscope is then inserted. Two additional 10 mm Ethicon Endopath Bladeless trocars separated by a hands’ breadth from the camera port are then placed under direct visualization.
7) Initially, the goal is to completely develop the anterior working space to the boundary of the inguinal ligament. The inguinal ligament can be identified anteriorly as a transverse structure comprised of white fibers. Using blunt dissection, the firm inguinal ligament can be palpated, confirming the extent of the dissection.
8) Identification of the adductor longus (medial boundary) and the sartorius (lateral boundary) muscles is then performed by identifying their overlying fascia as well as correlating the transillumination to the previously made skin markings. Once the above-mentioned landmarks have been identified, dissection begins.
9) Dissection of the fibrofatty lymph node tissue in the femoral triangle is begun using the Harmonic scalpel device. At the beginning of the dissection, the fascia lata is seen immediately. One must be careful not to inadvertently dissect deep into the fascia lata; this can be appreciated when reddish muscle fibers appear. With blunt dissection, the node packet can be rolled inwards on both sides to aid in resection. Small perforating vessels may be observed and bleeding can be controlled with the Harmonic scalpel.
10) Often, the saphenous vein can be visualized at the apex of the femoral triangle. If it is not visible, the entire posterior packet is dissected from where you can visualize the sartorious and adductor longus muscles coming together. While it is technically possible to dissect and spare the saphenous vein, we routinely divide it for oncologic reasons with a stapling device using a vascular load.
11) The fossa ovalis is then identified and the nodal packet is dissected away at its superolateral and superomedial limits from the inguinal ligament. The dissection is carried out deep to the fascia lata overlying the femoral vessels
12) Inferomedial dissection and skeletonization of the femoral vessels allows for resection of the deep inguinal nodes. This dissection should continue until the level of the femoral canal with visualization of the pectineus muscle to ensure complete nodal removal.
13) The nodal packet is then removed by means of an Endocatch. A 19 French Blake drain is placed through one of the working ports and the small skin incisions are closed, Figure 3a.
14) If a bilateral node dissection is planned, the same technique can be employed to the contralateral inguinal region.
Post-procedure instructionsTop
1) Antibiotics are continued until the patient is discharged from the hospital, which is almost invariably less than 24 hours.
2) The drain is kept in the dissection bed until drain output is noted to be less than 50 cc per day. This is
often for several weeks.
3) Both mechanical and chemical venous thromboembolism prophylaxis are given at
the operating surgeon’s discretion. Daily aspirin is preferred.
4) Patients are given a prescription for compression stockings and instructed to wear them immediately after surgery and when discharged. Referral to a lymphedema clinic is sometimes necessary to reinforce the importance of these devices. Patients otherwise have routine clinic follow up, Figure 3b.
Overall, 10 patients have been treated with 19 endoscopic groin dissections for penile cancer at our institution. Median nodal yield, operative time, hospital length of stay, and drain duration were 11 nodes/side (range 3-26), 177.5 minutes (range 132-400), 1 day (range 1-12 days), and 25 days (range 8-101 days), respectively. Six (31.6%) of the dissected groins to date have had positive lymph nodes (range = 1-2 positive nodes per positive side).
Initial complications data for patients undergoing endoscopic groin dissection for penile cancer have been encouraging. There were no perioperative mortalities. Major complications for patients undergoing groin dissection for penile cancer included readmission for incision and drainage (1 patient), pneumomediastinum without significant sequelae (1 patient), and readmission for superficial cellulitis requiring IV antibiotics
(1 patient). No patients have developed debilitating lymphedema, flap or skin edge necrosis, perioperative deep vein thrombosis or pulmonary embolism.
The most common minor complication was seroma formation in three patients. One patient developed mild-moderate lymphedema requiring daily compression stockings to help alleviate symptoms. One patient developed superficial cellulitis requiring oral antibiotics. Overall, using the Bevan-Thomas criteria, there were 7 complications (3 major, 4 minor) in 19 groin dissections (36.8%).
Local and distant recurrence occurred in 1 patient. Overall survival was 100% at 1 year post surgery. Currently, 7 of the 10 patients who underwent endoscopic groin dissection for penile cancer are alive. Mortalities for patients were attributed to recurrent metastatic disease (1 patient) and cardiovascular disease (2 patients).
Traditional open inguinal lymphadenectomy is the gold standard treatment for a variety of malignancies with nodal metastases when performed in a timely manner for clinically and pathologically appropriate patients. In a single institution study, early lymphadenectomy proved to be an independent prognostic factor for disease specific survival with a 3 year survival of 84% as compared to 35% for the delayed lymphadenectomy group.4 Additionally, patients with penile cancer that were managed with an extensive inguinal lymphadenectomy, defined as greater than eight nodes removed, experience an improved 5 year overall survival when compared to patients managed with a less extensive lymphadenectomy.5
Despite these advantages of early and extensive lymphadenectomy, open inguinal lymphadenectomy, and indeed, lymphadenctomy in general, remains an underutilized procedure. In the SEER (Surveillance, Epidemiology and End Results Program) database, only 26.5% of patients were treated with the recommended inguinal lymphadenectomy.5 This underutilization is often attributed to the substantial morbidity associated with this procedure. Even in centers where inguinal lymphadenectomy is routinely performed, there is a substantial rate of complications. Published cases have reported an overall wound complication rate ranging from 19%-51% and a wound dehiscence rate ranging from 7%-44% of open lymph node dissections.3 Furthermore, initial reports using the Bevan-Thomas complication system for morbidity of open lymphadenectomy reported a total complication rate of 57% in 106 dissections.8
These complications have driven several investigators to develop a minimally invasive approach to performing an inguinal node dissection6,9 to reduce morbidity while attempting to provide the same oncologic benefit. In our experience to date, the use of a laparoscopic technique has many advantages, namely, ease of adoption for an experienced laparoscopic surgeon, minimal learning curve, and improved complication profile, especially in obese patients. Using the same Bevan-Thomas system, patients in our study experienced 7 complications
(3 major, 4 minor) in 19 dissections (36.8%). In our small patient cohort, there was a reduced incidence of skin edge necrosis (0/19) and lymphedema (1/19), while the incidence of wound infection (3/19) and seroma (3/19) were comparable with an open approach.8
The short term oncologic data for performing endoscopic groin dissection for penile carcinoma is also promising. Patients in this series have had a 100% overall 1 year survival and 90% of the patients (9/10) have been disease free in short term follow up. These initial results are encouraging and hopefully will provide an oncologically equivalent alternative with an improved complication profile to the standard open inguinal lymph node dissection. In doing so, we postulate that patients who have pathology that would benefit from an inguinal node dissection will not defer surgery due to potential side effects.10
Institutional and population-based studies have shown that inguinal lymphadenectomies are not routinely performed in indicated patients. A minimally invasive approach to performing inguinal lymphadenectomy holds promise for providing a procedure with reduced morbidity and comparable oncologic benefit to the traditional open approach for patients with penile cancer. Further experience in a larger patient population with mature follow up is needed to validate this procedure in appropriate patients with high-risk penile cancer.
Accepted for publication June 2012
Address correspondence to Dr. Viraj A. Master, 1365 Clifton Road, NE, Building B, Suite 1400, Atlanta, GA 30322 USA
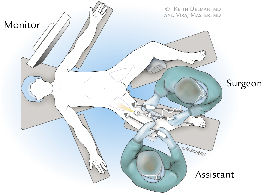
Figure 1a. Surgeon positioning in operating room.
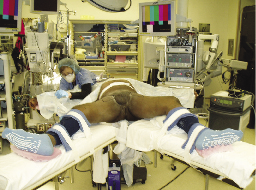
Figure 1b. Patient positioning with split-leg table.
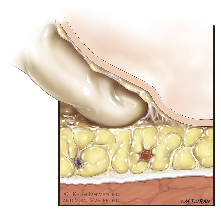
Figure 2. Blunt anterior dissection above Scarpa’s Fascia
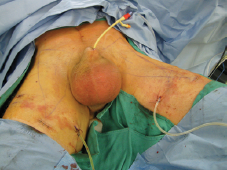
Figure 3a. Postoperative drain placement.
Figure 3b. Postoperative clinic follow up.
1. Siegel R, Naishadham D, Jemal A. Cancer statistics, 2012.
CA Cancer J Clin 2012;62(1):10-29.
2. Protzel C, Alcaraz A, Horenblas S, Pizzocaro G, Zlotta A, Hakenberg OW. Lymphadenectomy in the surgical management of penile cancer. Eur Urol 2009;55(5):1075-1088.
3. Horenblas S. Lymphadenectomy in penile cancer. Urol Clin North Am 2011;38(4):459-469, vi-vii.
4. Kroon BK, Horenblas S, Lont AP, Tanis PJ, Gallee MP, Nieweg OE.
Patients with penile carcinoma benefit from immediate resection of clinically occult lymph node metastases. J Urol 2005;
173(3):816-819.
5. Johnson TV, Hsiao W, Delman KA, Jani AB, Brawley OW, Master VA. Extensive inguinal lymphadenectomy improves overall 5-year survival in penile cancer patients: results from the Surveillance, Epidemiology, and End Results program. Cancer 2010;116(12):2960-2966.
6. Master VA, Ogan K, Kooby D, Hsiao W, Delman K. Leg endoscopic groin lymphadenectomy (LEG procedure): step-by-step approach to a straightforward technique. Eur Urol 2009;56(5):
821-828.
7. Dindo D, Demartines N, Clavien PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 2004;240(2):205-213.
8. Bevan-Thomas R, Slaton JW, Pettaway CA. Contemporary morbidity from lymphadenectomy for penile squamous cell carcinoma: the M.D. Anderson Cancer Center Experience. J Urol
2002;167(4):1638-1642.
9. Tobias-Machado M, Tavares A, Molina WR Jr, Forseto PH Jr, Juliano RV, Wroclawski ER. Video endoscopic inguinal lymphadenectomy (VEIL): minimally invasive resection of inguinal lymph nodes. Int Braz J Urol 2006;32(3):316-321.
10. Master VA, Jafri SM, Moses KA, Ogan K, Kooby DA, Delman KA.
Minimally invasive inguinal lymphadenectomy via endoscopic groin dissection: comprehensive assessment of immediate and long-term complications. J Urol 2012;In Press.
